Explain the concept of Hydrolysis.
In its simplest definition, hydrolysis is a chemical reaction in which water is used to break down the bonds of a particular substance. In biotechnology and as far as living organisms are concerned, these substances are often polymers.
The word hydrolysis comes from the word hydro, which is Greek for water, and lysis, which means “to unbind.” In practical terms, hydrolysis implies the act of separating chemicals when water is added. There are three main types of hydrolysis: salt, acid, and base hydrolysis.
Hydrolysis can also be thought of as the exact opposite reaction to condensation, which is the process whereby two molecules combine to form one larger molecule. The end result of this reaction is that the larger molecule ejects a water molecule.
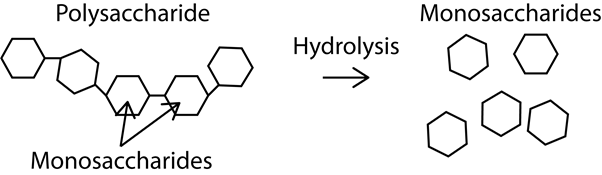
Three common types of hydrolysis
Salts: Hydrolysis occurs when salt from a weak base or acid dissolves in liquid. When this happens, water spontaneously ionizes into hydroxide anions and hydronium cations. This is the most common type of hydrolysis.
Acid: Water can act as an acid or a base, according to the Bronsted-Lowry acid theory. In this case, the water molecule would give away a proton. Perhaps the oldest commercially-practised example of this type of hydrolysis is saponification, the formation of soap.
Base: This reaction is very similar to the hydrolysis for base dissociation. Again, on a practical note, a base that often dissociates in water is ammonia.[/vc_column_text][/vc_column][/vc_row]
Tag:hydrolysis, polymers